Sequencing, spatial biology, and PCR platforms all rely on microfluidic devices such as flow cells, microfluidic chips, and test cartridges. In these systems, how precisely and reliably fluids move through the device has a direct impact on run quality, instrument uptime, and ultimately, system costs.
While precise flow control is critical, many instrument designers overlook a significant opportunity to improve performance and reduce costs. The pumps and valves that enable the flow path are often selected based on legacy architectures. By optimizing component selection and integration strategy, you can uncover opportunities to improve assay performance, simplify manufacturability, and reduce service and warranty costs.
In this article, we’ll explore the challenges designers face when moving fluid to and through a flow cell – and highlight pump and valve solutions that address these issues while improving cost efficiency, reliability, and operational precision in modern microfluidic instruments.
While there are many ways to reduce overall system costs, fluid-handling introduces a distinct set of constraints that are often more difficult to address.
Specifically, design engineers face several core fluidic challenges when directing fluid to a microfluidic chip or flow cell. Successfully addressing these challenges requires approaches that:
These challenges have historically locked designers into relying on traditional methods such as syringe pumps and rotary valves, which are large, expensive, and costly to maintain.
Figure 1 shows a common direct liquid handling architecture in use today.

Figure 1. Schematic showing a syringe pump pushing reagents/wash buffers through the flow cell.
This architecture is widespread in DNA/RNA sequencing, spatial biology, and other applications where extremely low internal volume and minimal dead volume are non-negotiable. However, it tends to come with high maintenance requirements that can significantly add to operating costs over time.
Alternative options do exist.
Piston pumps and solenoid valves offer compelling cost advantages. Figure 2 presents an alternative architecture that leverages these components to address many of the cost and maintenance challenges associated with traditional systems.

Figure 2. Schematic showing a manifold with LFN solenoid valves, an Xover® solenoid valve, and an LPD variable volume pump.
This design replaces the rotary shear valve with a selector manifold equipped with individual LFN Series and Xover® solenoid valves. In this scenario, reagents A, B, and C are selected using an LFN manifold. Because the last two reagents are more expensive, they are controlled separately by the Xover – a 3‑way isolation solenoid valve specifically optimized for low internal and carryover volume. Beyond reagent selection, the Xover can also be used to choose between flow cells or for diverting or recirculating flow.
An LPD Series piston pump is positioned downstream of the flow cell to pull fluid through the flow path. An LFV solenoid valve serves as both the inlet and outlet valve for the pump. The inlet valve allows aspiration of fluid into the pump through the flow cell, while the outlet valve switches states to dispense the fluid to waste.
Each approach comes with unique tradeoffs – the right choice depends on which constraints matter most for your application.
The comparison table below shows how our components measure up across key performance metrics.
| Components | Benefits | Considerations |
| LPD Piston Pump & LFV Solenoid Valve | LPD:
LFV:
|
|
| LFN Selector Multi‑Valve Manifold |
|
|
| Xover® Solenoid Valve |
|
|
While rotary shear valves remain the benchmark for minimizing dead volume, our team works closely with customers to optimize manifold flushability and overall fluidic performance.
Choosing components with a higher cycle life reduces maintenance frequency and replacement costs, minimizing instrument downtime and lowering total cost of ownership. Our pumps and valves are engineered for long service life and typically operate maintenance‑free –often at a fraction of the cost of traditional syringe pumps and rotary shear valves.
Precise fluid delivery ensures reproducible results across runs, which is critical for maintaining data quality and assay reliability.
Figures 3 and 4 show how the LPD Series pump demonstrates consistent dispensing precision and accuracy across life on DMSO.

Figure 3. Graph depicting CV of the LPD Series Pump at 100% dispense over life on DMSO.
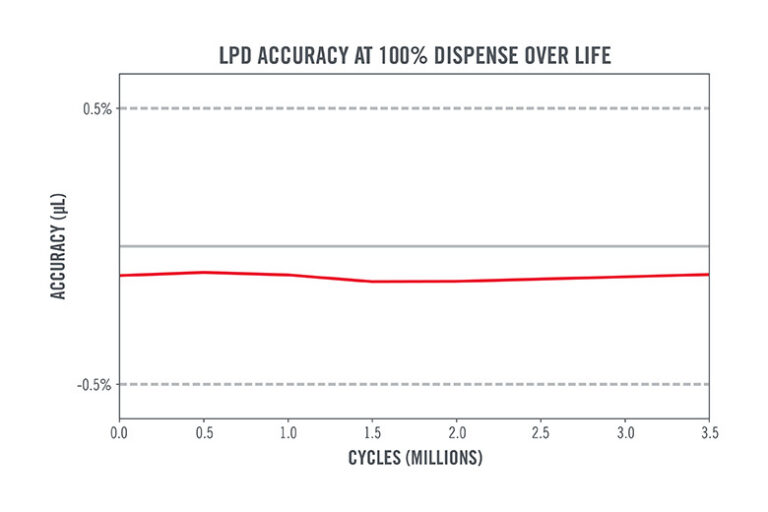
Figure 4. Graph depicting dispense accuracy of LPD Series Pump over life on DMSO.
Poor flushability leads to cross-contamination between reagents, causing unintended reactions that can degrade performance in downstream processes. The Xover helps to minimize cross-contamination and reduces flushing time. This increases sample throughput while lowering reagent consumption.
Figure 5 compares Xover flushability against that of two traditional rocker-style solenoid valves. Xover required 32% less volume than the next rocker valve to flush from 10 PPM to 1 PPM.

Figure 5. Graph comparing flushability of the Xover against that of two traditional rocker-style solenoid valves, where Xover required 32% less volume than the next rocker valve to flush from 10 PPM to 1 PPM.
Syringe pumps, peristaltic pumps, and many other positive displacement pumps have fundamental tradeoffs: they’re non-continuous, respond slowly to setpoint changes, and can produce pulsatile, unstable flow.

Figure 6. Schematic showing non-contact dispenses onto a flow cell using a pipette module.
Figure 6 illustrates a traditional approach to non-contact dispensing onto a flow cell using a bidirectional pump and a pipette. The pipette tip introduces its own limitations – dispensing accuracy can be inconsistent and the tip may require disposal or washing between samples, adding costs and additional user touchpoints.
As an alternative, some instrument designers use traditional pressure-driven flow systems to move fluid to a microfluidic chip or flow cell. However, this typically requires an air pump and pressure controller which are historically large and induce pulsatility.
To solve this challenge, we’ve created multiple pressure-driven flow solutions to move fluid to and through a flow cell with our miniature disc pumps and HDI solenoid valves. Figure 7 demonstrates how these components can be used to pipette fluid to a flow cell, as well as push and pull fluid through a flow cell or microfluidic chip using this pneumatic approach. Note, the disc pumps and HDI solenoid valves do not flow liquid directly, but rather use pressure-driven flow to drive the liquids downstream.

Figure 7. Schematic showing a disc pump used for non-contact dispensing/pipetting into the flow cell and on-chip flow reversal (pushing and pulling fluid) through the flow cell, then diverting to waste via a 3-way Xover solenoid valve.
This compact architecture delivers the slow, steady, laminar flow needed in microfluidics, with rapid response times and highly controllable pressure settings in a reliable, cost-effective package:
Our disc pump enables ultra-smooth, low shear, laminar flow streams ideal for microfluidic devices in sequencing, spatial biology, PCR, and organ-on-a-chip (OoC). The disc pump can generate pressure and vacuum, and two HDI pneumatic solenoid valves can be packaged with it on a manifold to switch between the two, reversing flow direction with ease. This allows pulsation-free pushing and pulling of reagents through a flow cell using pneumatics.

Figure 8. The disc pump provides ultra-smooth, low shear, laminar flow. This graph compares the flow rate stability output of a conventional syringe pump versus the disc pump.
Since the disc pump is a compact and continuous pump, it can also be used to address multiple flow cell lanes at once – enabling multiplexed architectures with a few additional solenoid valves.
With the use of a pressure and flow sensor, it also enables the monitoring of fluid resistance on-chip, providing insights into the biological and mechanical changes happening inside the chip – like pressure spikes from bubbles or clogging. Because of the disc pump’s rapid set point response and pulsation-free flow, it can maintain specific pressure/flow profiles, even as changes to fluid restriction take place. This helps to maintain sample and chip integrity and avoid unnecessary waste, all without the limitation of stroke/refilling of alternative pressure sources (like a syringe pump). These real-time adjustments to fluid resistance may salvage instrument runs and optimize efficiencies of on-chip reactions, reducing waste and downtime.
Since the disc pump is not a flow-through device, the Xover can also be used to divert fluid to waste.
>>Application Notes AN002 and AN007 discuss flow reversal in microfluidics.
A pneumatic approach using disc pump and HDI pneumatic solenoid valves can also be used to pipette fluid into the flow cell and dispense to waste. This presents a very compact, lightweight, and cost effective pipettor that removes the need for bulkier, pulsatile air pumps and pressure controllers, and places the pump directly above the dispense tip on a fast-moving robotic arm. The pump’s reduced size and weight directly enable faster arm movement, improving overall throughput. In direct liquid handling architectures, it is also possible to accomplish this using our LPD Series piston pump.
>>In Application Note AN049, we showcase how you can use a Smart Pump Module (SPM) as a volume control module to design a compact, lightweight pipettor. Combined with an integrated sensor and electronics, this is an all-in-one pressure-regulator solution that eliminates the need for other system components.
We’ve spent over 75 years helping engineers like you navigate complex fluidic challenges throughout every step of the diagnostic process – from sample preparation to amplification, detection, and sequencing. Chances are, we already have the precise flow control solution you need. If not, our engineers can modify our COTS components to fit your specific requirements without significantly impacting cost or lead times.
Lee engineers specialize in working with customers on an engineer-to-engineer level to navigate design and material complexities with confidence. Our global presence allows us to deliver local technical support that keeps development moving and innovation on track. Every Lee component is 100% functionally tested to ensure performance throughout your system’s lifecycle. Together, these strengths accelerate your path to market with components you can rely on.
Let us help you mitigate risk, optimize performance, and bring your medical innovations to life. Connect with a Lee Sales Engineer today to discuss your diagnostic application.
Always verify flow calculations by experiment.
*There are many parameters to consider when determining V-Factor. Click here for more information.