Bulk modulus is a measure of the resistance of a fluid to compression. It is defined as the ratio of pressure stress to volumetric strain. Assuming there is no entrained air in the system, bulk modulus can be expressed using the following formula:
Where:
B = Bulk modulus (psi)
∆P = Change in pressure (psi)
∆V = Change in volume
V = Initial volume
When the value of B is known (see reference table below), it is easy to calculate the effect of any pressure change on volume, or of any volume change on pressure.
EXAMPLE:
MIL-H-83282 oil has a bulk modulus of 3.0 x 105 psi. Thus, a pressure increase of 3000 psi will reduce its volume by 1.0%.
Thermal expansion is the tendency of a fluid to expand due to an increase in temperature. The coefficient of thermal expansion can be used to relate temperature change to volume change, as shown in the following formula.
Where:
γ = Coefficient of cubical thermal expansion/°F at 100°F
∆T = Temperature rise, °F
EXAMPLE:
MIL-H-83282 oil has a coefficient of cubical thermal expansion of 0.00046/°F. Thus a temperature rise of 100°F will increase a 2.0 gallon volume by 0.09 gallons.
The bulk modulus and the coefficient of cubical thermal expansion can be used together to compute the pressure rise in a closed system subjected to an increasing temperature.
EXAMPLE:
MIL-H-83282 oil at 0 psi is heated from 70°F to 120°F in a closed, constant volume system containing 100 cubic inches.
This is the same ∆P that would be caused by adding 2.3 cubic inches of oil with no temperature change. It is also apparent that a constant system pressure could be maintained by bleeding off 2.3 cubic inches of oil while increasing the temperature by 50°F.
REFERENCE TABLE
| FLUID | Bref. | γ |
| Units | psi | ΔV/V/°F |
|---|---|---|
| Gasoline | 150 000 | 0.00072 |
| JP-4 | 200 000 | 0.00057 |
| MIL-H-5606 | 260 000 | 0.00046 |
| MIL-H-83282 | 300 000 | 0.00046 |
| MIL-H-6083 | 260 000 | 0.00044 |
| SKYDROL 500B-4 | 340 000 | 0.00047 |
| Silicone 100cs | 150 000 | 0.00054 |
| Water | 310 000 | 0.00021 |
Where:
∆V = Change in volume
V = Initial volume
∆T = Temperature rise, °F
Bref. = Tangent adiabatic bulk modulus psi stated at 100°F, 2500 psi and no entrained air. A reference point.
γ = Coefficient of cubical thermal expansion/°F at 100°F
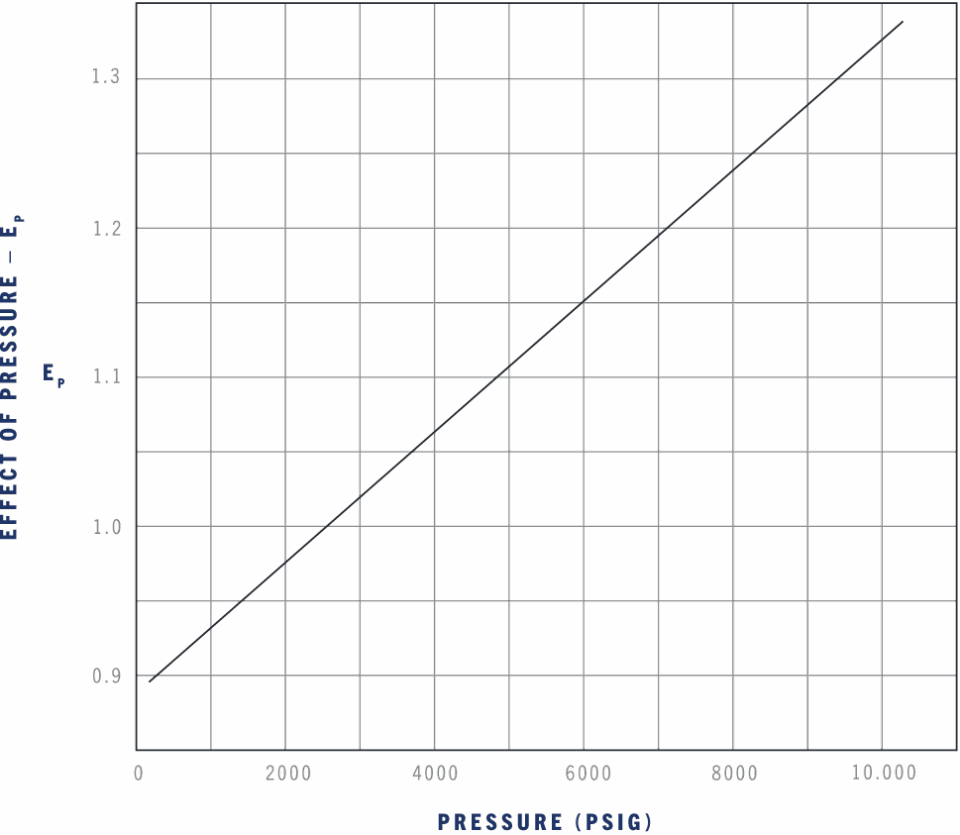
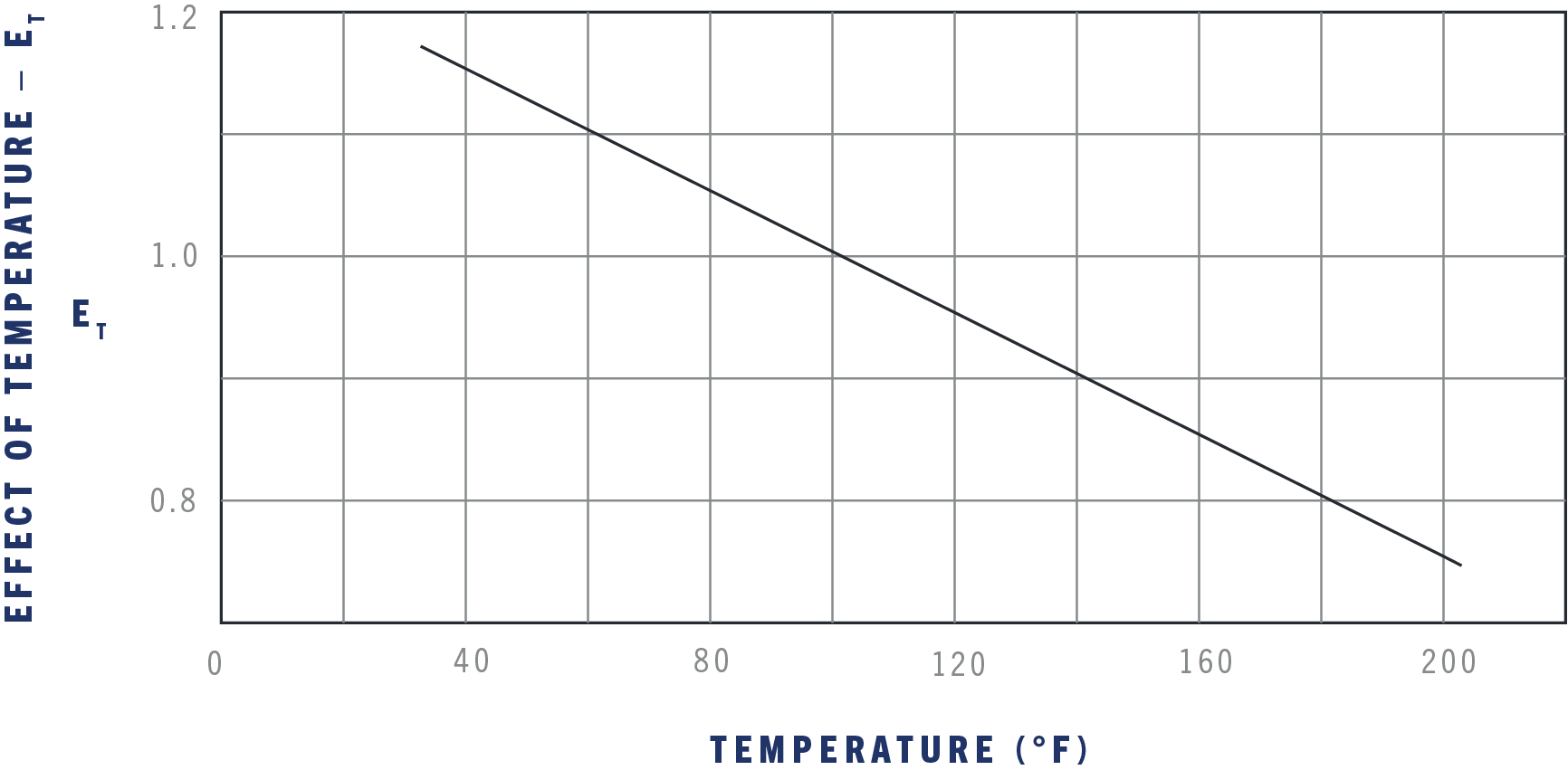
The previous examples used a constant bulk modulus for simplicity. In actual use, the bulk modulus is affected by the working pressure, temperature, and percent of entrained air. Use the next three graphs to find the effect of these variables and you will get a close approximation of actual conditions. The actual bulk modulus, B, of a fluid is the value in the table as Bref. modified for the effect of pressure, temperature, and percent of entrained air.
Where:
EP = Effect on pressure
ET = Effect on temperature
EA = Effect on entrained air
EXAMPLE:
500 psi, 60°F, 2% entrained air, MIL-H-83282.
EXAMPLE:
2000 psi, 160°F, 2% entrained air, MIL-H-83282.
With the corrected bulk moduli for the two end points of a thermal problem, an average bulk modulus can be selected for calculation purposes. We would use 244,000 psi for B.
The effect of working pressure on bulk modulus for hydrocarbon fluids.
The effect of temperature on bulk modulus for hydrocarbon fluids.
The effect of entrained air on bulk modulus in hydrocarbon and other fluids for different working pressures.
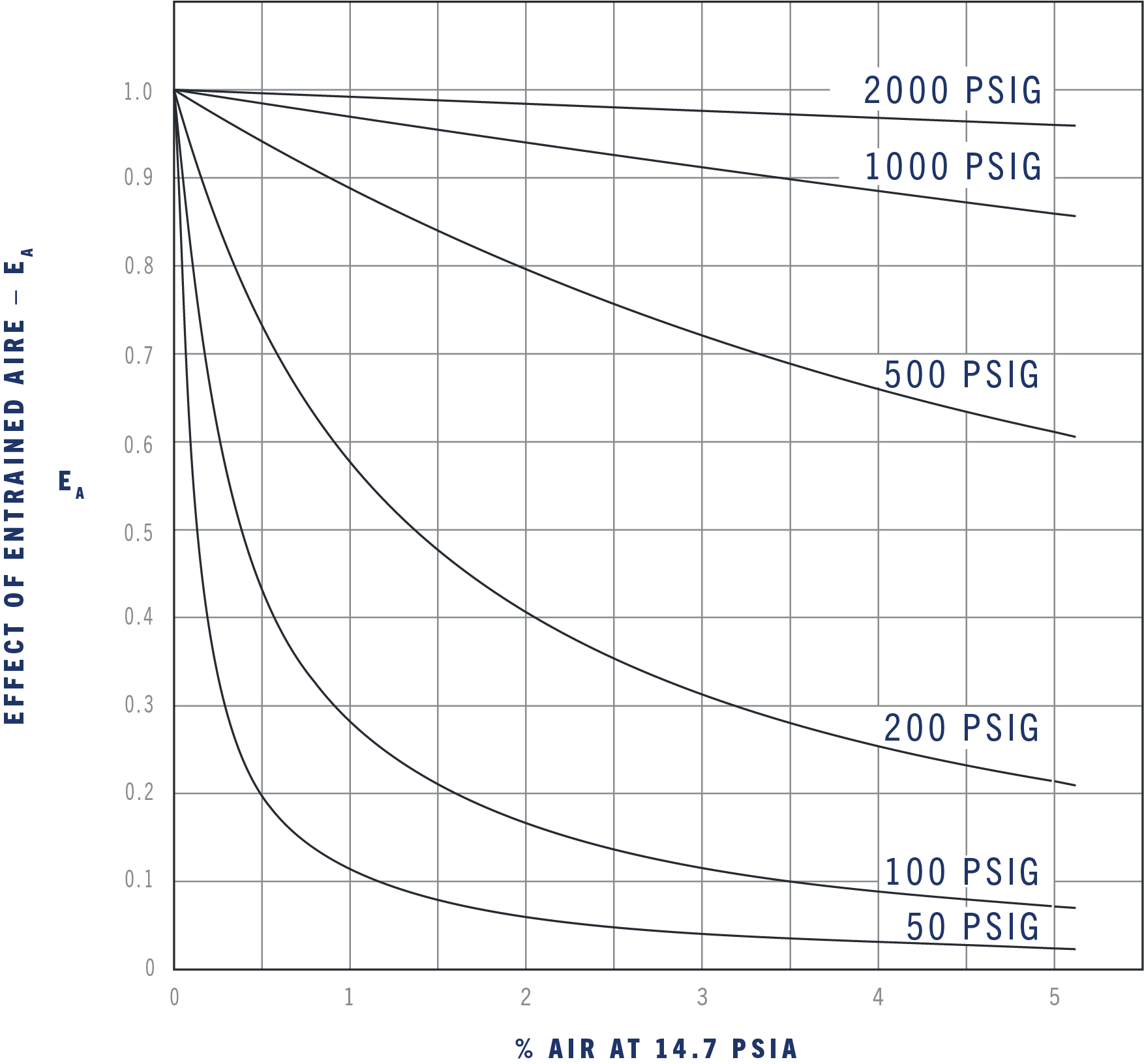
To simplify the calculations of thermal problems with entrained air, these curves show the average effect on a 230,000 psi bulk modulus for pressure points fairly close together. If a wide change in pressure is encountered in a problem, it would be more accurate to break the changes down into two or more steps, depending on the accuracy desired.
An accurate one-step formula for this relationship follows:
(Note that pressure is in units of psia.)
Always verify flow calculations by experiment.
*There are many parameters to consider when determining V-Factor. Click here for more information.